The Paper of the Month November
01 Nov 2021A new WSA feature highlighting original research in the field of neurology, selected by WSA Associate Commissioning Editor, Anita Arsovska
Editorial
Title: Is dual antiplatelet therapy with cilostazol a new therapeutic option for stroke prevention in patients with intracranial stenosis?
Author: Prof. Anita Arsovska, MD, PhD, FESO
This article is a commentary on the following
Summary:
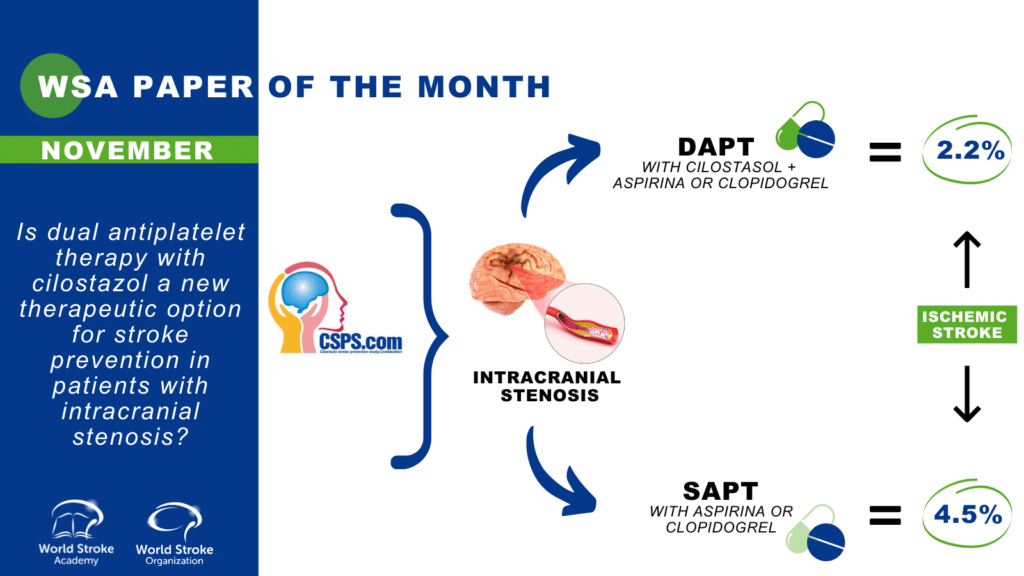
The authors performed a subgroup analysis of the Cilostazol Stroke Prevention Study for Antiplatelet Combination (CSPS.com) trial, a randomized controlled trial in high‐risk Japanese patients with ischemic stroke aiming to compare the efficacy and safety of dual antiplatelet therapy (DAPT) with cilostazol and clopidogrel or aspirin to those of single antiplatelet therapy (SAPT) with clopidogrel or aspirin in patients with intracranial symptomatic or asymptomatic arterial stenosis (ICAS) of at least 50%¹.
Patients were allocated to receive either DAPT (n=275) or SAPT (n=272) The risks of ischemic stroke (hazard ratio [HR], 0.47; 95% CI, 0.23–0.95); and composite of stroke, myocardial infarction, and vascular death (HR, 0.48; 95% CI, 0.26–0.91) were lower in DAPT than SAPT, whereas the risk of severe or life‐threatening bleeding (HR, 0.72; 95% CI, 0.12–4.30) did not differ between the 2 treatment groups.
DAPT using cilostazol was superior to SAPT (either clopidogrel or aspirin) for the prevention of recurrent stroke and vascular events without increasing bleeding risk among patients with intracranial arterial stenosis after stroke.
Commentary:
DAPT after ischemic stroke or transient ischemic attack may reduce recurrent stroke but also increase severe bleeding compared with SAPT.
The CSPS.com trial Investigators reported that, among 1879 Japanese patients with high-risk noncardioembolic ischemic stroke in the previous 8 to 180 days, adding cilostazol to single antiplatelet therapy with aspirin or clopidogrel reduced the annual incidence of recurrent ischemic stroke by half compared with monotherapy (2.2% dual therapy with cilostazol versus 4.5% monotherapy, hazard ratio, 0.49 [95% CI, 0.31–0.76]), without increasing the annual risk of severe or life-threatening bleeding (0.6% dual versus 0.9% monotherapy, hazard ratio, 0.66 [0.27–1.60])².
Neurologists are always looking for new possibilities to improve stroke prevention, so can these results affect our usual clinical treatment strategy?
Hankey GJ³ analysed the results of the CSPS.com trial regarding bias (detection bias, performance bias, attrition bias), precision, plausibility, consistency and generalizability. Most important points were regarding precision and generalizability. The precision analysis showed wide range of recruitment (13-62 days, median 26 days), with short follow-up period (median 1.4 years), quite small total number of primary outcome events (recurrent ischemic stroke) in each group (29 [3%] for DAPT and 64 [7%] for SAPT), wide 95% CI of the hazard ratio of recurrent ischemic stroke with DAPT vs SAPT (0.31–0.76).Also, the estimate of the hazard ratio of severe or life-threatening bleeding was imprecise, ranging from a 73% reduction to a 60% increase with DAPT vs SAPT. Regaring generalizability, all of the RCTs of cilostazol in patients with ischemic stroke have been conducted in Asian countries and it remains uncertain whether the results of these trials are generalizable to other populations in other continents.
The recent review on the American Heart Association/American Stroke Association Guidelines 2021² concluded that DAPT (aspirin plus clopidogrel) was more effective than SAPT for prevention of secondary ischemic stroke when initiated early after the onset of minor stroke/high-risk transient ischemic attack and treatment duration was <90 days. When the treatment duration was longer and initiated later after stroke or transient ischemic attack onset, DAPT was not more effective than SAPT for ischemic stroke prevention and it increased the risk of bleeding². However, trials regarding intracranial stenosis/use of cilostazol were not included in this meta-analysis due to insufficient/heterogenous data⁴.
Another recent review and meta-analysis aimed to determine the efficacy and safety of cilostazol for secondary stroke prevention³. It included 18 randomised trials with 11 429 participants. There were 3 studies comprising 755 patients that included only patients with symptomatic ICAS. For most outcomes, there was no significant difference between the symptomatic ICAS and mixed/other stroke subgroup. In the ICAS subgroup, cilostazol administration was associated with an insignificant increase in risk of MACE (RR=1.38, 95% CI 0.67 to 2.85, p=0.38), compared with a statistically significant decrease in MACE risk in studies which did not exclusively recruit patients with symptomatic ICAS. There was no significant decrease in risk of ICAS progression or worsening (RR=0.63, 95% CI 0.25 to 1.58, p=0.33) after cilostazol administration⁵.
Cilostazol use in patients who have contraindications (resistance or higher risk for haemorrhagic complications) to Aspirin or Clopidogrel?
Cilostazol selectively inhibits phosphodiesterase 4 and adenosine reuptake, increases activation of intracellular cyclic-AMP, inhibits platelet aggregation and dilates the blood vessels. The analysis by Hankey GJ³ of the CSPS trial suggests that cilostazol took several months to take effect and alter stroke risk and hypothesized that that the vasodilatory (and other pleiotropic) properties of cilostazol may be as, or more important, than any antiplatelet properties.
A nation-wide cohort study by Lee TH et al.⁶ compared the long-term efficacy and safety between cilostazol and clopidogrel in chronic ischemic stroke and showed a trend of lower frequency of recurrent acute ischemic stroke or major bleeding in patients with a history of hypertension or gastrointestinal bleeding in the cilostazol group.
A recent subgroup analysis of the PICASSO Trial⁷ compared the efficacy and safety of cilostazol vs aspirin in ischemic stroke patients with high-risk cerebral hemorrhage and found that hemorrhagic stroke was less frequent in the cilostazol than in the aspirin group in patients with multiple microbleeds, concluding that Cilostazol may be more beneficial for ischemic stroke patients with multiple cerebral microbleeds and before white matter changes are extensive.
Conclusion: Although DAPT with cilostazol has shown efficacy and safety in secondary stroke prevention in patients with intracranial stenosis, further randomized clinical trials conducted outside Asia are needed to increase the generalizability to populations with a different ethnic background (or non-Asiatic populations).
References
- Uchiyama S, Toyoda K, Omae K et al. Dual Antiplatelet Therapy Using Cilostazol in Patients With Stroke and Intracranial Arterial Stenosis. Journal of the American Heart Association. 2021;0:e022575. https://doi.org/10.1161/JAHA.121.022575
- Toyoda K, Uchiyama S, Yamaguchi T, Easton JD, Kimura K, Hoshino H, Sakai N, Okada Y, Tanaka K, Origasa H, Naritomi H, Houkin K, Yamaguchi K, Isobe M, Minematsu K; CSPS.com Trial Investigators. Dual antiplatelet therapy using cilostazol for secondary prevention in patients with high-risk ischaemic stroke in Japan: a multicentre, open-label, randomised controlled trial. Lancet Neurol. 2019 Jun;18(6):539-548. doi: 10.1016/S1474-4422(19)30148-6. PMID: 31122494.
- Hankey GJ. CSPS.com Trial of Adding Cilostazol to Antiplatelet Therapy to Reduce Recurrent Stroke. 2020;51:696–698https://doi.org/10.1161/STROKEAHA.119.028409
- Devin L. Brown DL, Levine DA, Albright K et al. Benefits and Risks of Dual Versus Single Antiplatelet Therapy for Secondary Stroke Prevention: A Systematic Review for the 2021 Guideline for the Prevention of Stroke in Patients With Stroke and Transient Ischemic Attack. 2021;52:e468–e479. https://doi.org/10.1161/STR.0000000000000377
- Tan CH, Wu AG, Sia CH, et al. Cilostazol for secondary stroke prevention: systematic review and meta-analysis. Stroke Vasc Neurol. 2021;6(3):410-423. doi:10.1136/svn-2020-000737
- Lee T-H, Lin Y-S, Liou C-W, Lee J-D, Peng T-I, Liu C-H. Comparison of long-term efficacy and safety between cilostazol and clopidogrel in chronic ischemic stroke: a nationwide cohort study. Therapeutic Advances in Chronic Disease. January 2020. doi:1177/2040622320936418
- Kim BJ, Kwon SU, Park JH et al.; PICASSO Investigators. Cilostazol Versus Aspirin in Ischemic Stroke Patients With High-Risk Cerebral Hemorrhage: Subgroup Analysis of the PICASSO Trial. Stroke. 2020 Mar;51(3):931-937. doi: 10.1161/STROKEAHA.119.023855. Epub 2019 Dec 20. PMID: 31856691.
Author Interview
Prof. Shinichiro Uchiyama, M.D., Ph.D.
1. WHAT DID YOU SET OUT TO STUDY?
This was a subgroup analysis of the CSPS.com trial, which was a randomized controlled trial to compare dual antiplatelet therapy (DAPT) with cilostazol and aspirin or clopidogrel to single antiplatelet therapy (SAPT) with aspirin or clopidogrel in high-risk Japanese patients with non-cardioembolic ischemic stroke in order to investigate superiority of DAPT to SAPT for the long-term prevention of recurrent stroke among patients with intracranial arterial stenosis (ICAS).
2. WHY THIS TOPIC?
Long-term benefit of DAPT over SAPT for the prevention of recurrent stroke has not been established in patients with ICAS. Based on the results of WASID and SAMPRIS trials, the 2021 version of ASA/AHA guidelines recommend short-term DAPT plus aggressive risk factor management in patients with severe ICAS in the vascular territory of ischemic stroke or TIA. However, according to a meta-analysis of aspirin plus clopidogrel versus aspirin alone for the prevention of recurrent stroke in patients with minor ischemic stroke or high-risk TIA, including the CHANCE and POINT trials, risk of recurrent stroke was reduced by DAPT only for a few weeks, and risk of bleeding increased until 3 months. In addition, clopidogrel resistance is more common in East Asians including Japanese. In reality, according to a genetic analysis of the CHANCE trial, the use of clopidogrel plus aspirin, as compared with aspirin alone, the risk of recurrent stroke reduced only in the subgroup of patients without clopidogrel resistance. The results of the CSPS.com trial showed that recurrent stroke and vascular events were significantly fewer in the DAPT group than in the SAPT group, whereas serious or life-threatening bleeding was comparable between both groups. Based on this background, we conducted a subgroup analysis of ICAS in patients recruited in the CSPS.com trial to compare DAPT with cilostazol and aspirin or clopidogrel and SAPT with aspirin or clopidogrel in high-risk patients with ischemic stroke.
3. WHAT WERE THE KEY FINDINGS?
Dual antiplatelet therapy with cilostazol was superior to aspirin or clopidogrel alone for the long-term prevention of recurrent stroke and vascular events without increasing bleeding risk in patients with ischemic stroke and ICAS.
4. HOW MIGHT THESE RESULTS IMPACT CLINICAL PRACTICE?
This was the first evidence to support that DAPT with cilostazol can be a therapeutic option for the long-term prevention of recurrent stroke and vascular events without increased bleeding in patients with symptomatic or asymptomatic ICAS of at least 50% after ischemic stroke.
5. WHAT SURPRISED YOU MOST?
It was unclear that DAPT with cilostazol is superior to aspirin or clopidogrel alone since the risk of recurrent stroke is still high despite adherence to the current guidelines including DAPT with aspirin and clopidogrel plus intensive risk factor management in patients with symptomatic ICAS. In this CSPS.com subgroup analysis, the risk of stroke, ischemic stroke, and vascular events were less than half in DAPT with cilostazol than aspirin or clopidogrel alone, whereas the bleeding risk did not differ.
6. WHAT’S NEXT FOR THIS RESEARCH?
This study has some limitations. First, the sample size was relatively small and the evidence level was limited because of a subgroup analysis. Second, it remains uncertain whether the present results can be generalized to other ethnicities than Japanese. Third, one cannot tell from these data whether DAPT is preventing recurrent stroke related to ICAS or just preventing stroke in any territory related to other mechanisms of stroke in patients with asymptomatic ICAS. Therefore, a large randomized controlled trial of DAPT with cilostazol versus aspirin or clopidogrel alone for the long-term prevention of recurrent stroke is required in multiethnic patients with symptomatic ICAS.
7. IS THERE ANYTHING YOU’D LIKE TO ADD?
The CSPS.com trial did not include patients within 7 days after stroke. Another subgroup analysis of the CSPS.com trial suggests that long-term dual antiplatelet therapy using cilostazol was more effective for secondary stroke prevention than monotherapy in patients initiating the medication at 15 days or later. Therefore, DAPT with aspirin and clopidogrel within a few weeks after stroke, followed by DAPT with cilostazol and aspirin or clopidogrel thereafter, could be a strategy for the short-term to long-term prevention of recurrent stroke without increasing bleeding risk in ICAS patients.